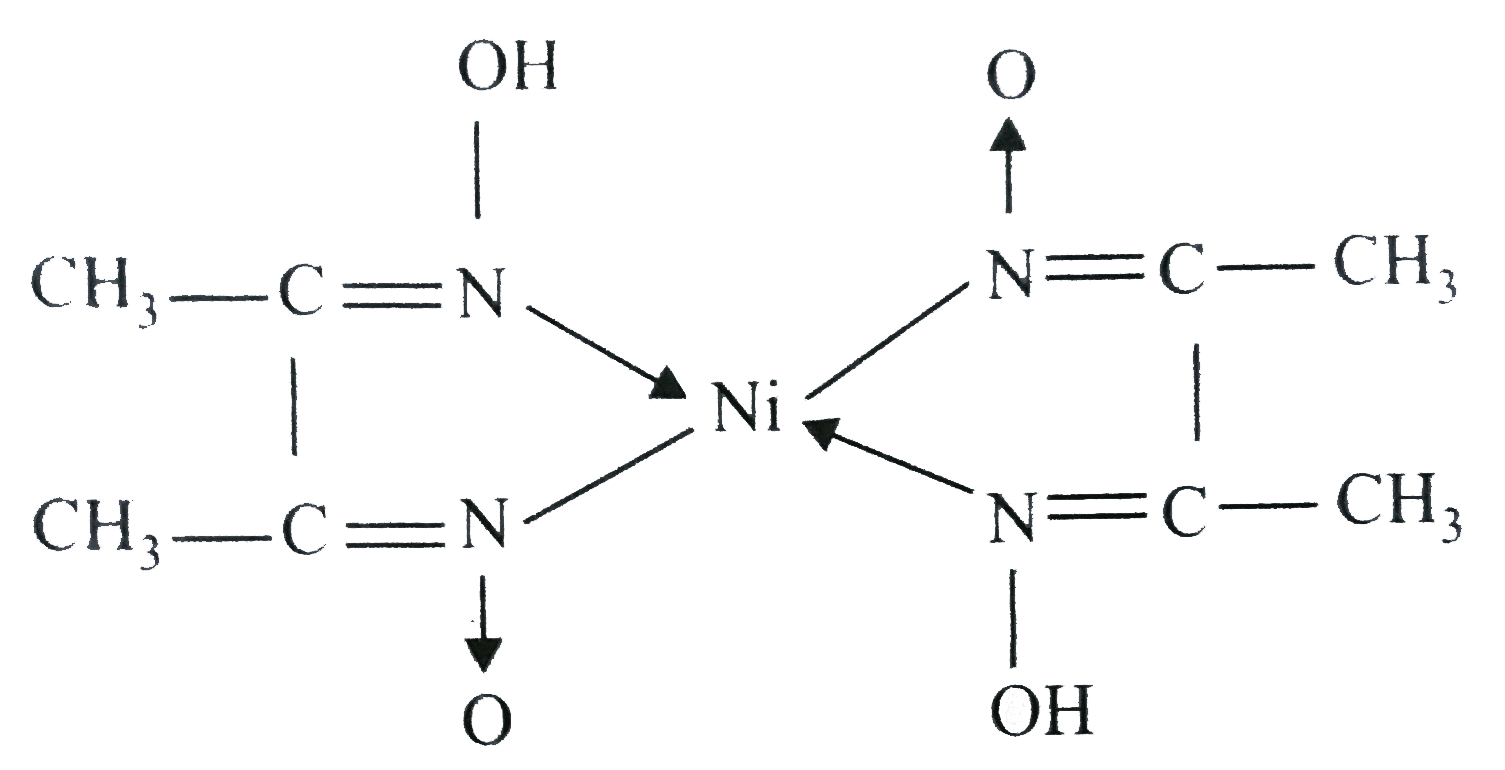
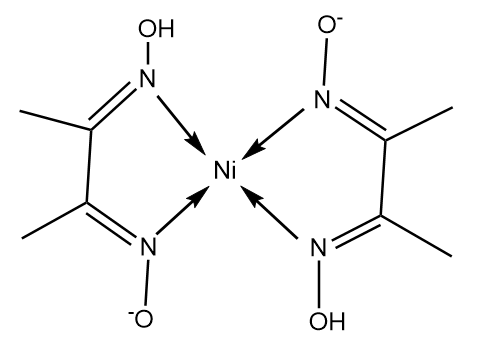
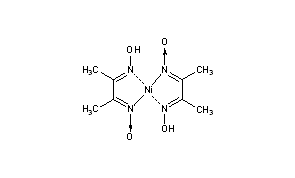
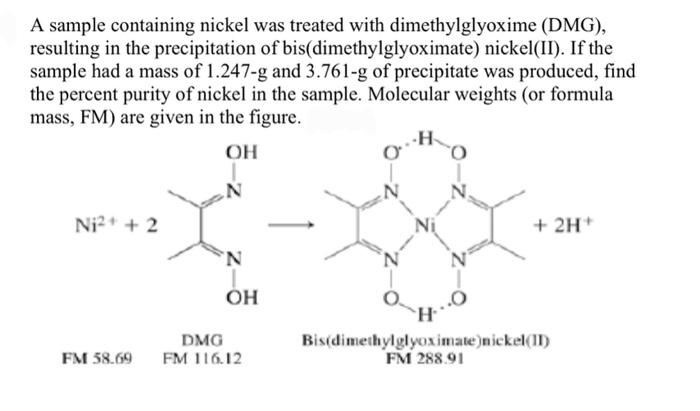
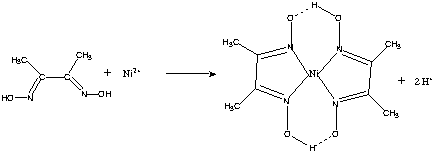
In basic medium the amount of Ni2+ in a solution can be estimated with the dimethylglyoxime reagent. The correct statement(s) about the reaction and the product is(are):

Dimethylglyoxime (C4H8N2O2) - Structure, Molecular Mass, Properties and Uses of Dimethylglyoxime, Dimethylglyoximato Ligand

Ultratrace Detection of Nickel(II) Ions in Water Samples Using Dimethylglyoxime-Doped GQDs as the Induced Metal Complex Nanoparticles by a Resonance Light Scattering Sensor | ACS Omega
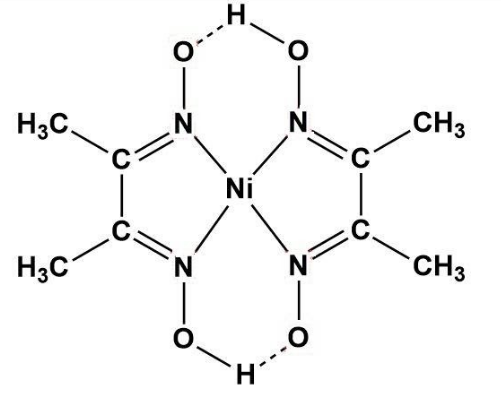
An alcoholic solution of dimethylglyoxime is added to an aqueous solution of nickel (II) chloride. Slow addition of ammonium hydroxide leads to the precipitation of a rosy red coloured metal complex. Then








![The IUPAC name of the complex Ni[C(4)H(7)O(2)N(2)] formed by the react The IUPAC name of the complex Ni[C(4)H(7)O(2)N(2)] formed by the react](https://d10lpgp6xz60nq.cloudfront.net/physics_images/RS_P2_CHM_C09_E01_015_S01.png)